Treasury Department: To close out the #G20 Finance Meetings, @SecYellen will give a press conference to discuss global tax policy, clima… https://t.co/ .... Find data and reports related to the Paycheck Protection Program (PPP). Learn more · Get help with your PPP loan. SBA offers free counseling and training events ...
SOP 101: Interim Monitoring Visits and Closeout Visit. Version # 4.0 ... Reaction. • AE Reports: Investigator reports of all adverse events (both serious sand non-.. Jul 18, 2016 — The closeout visit report signifies closure of investigational sites with submission and signature. The monitoring visit report is the full .... Close. State of Nevada Seal Department of Health and Human Services Division of ... This outside entity that has “spoofed” the SNAP toll free information hotline ... If you have already fallen victim to this or a similar scam, please visit: FTC Identity Theft Information. Important Links. REPORT WELFARE FRAUD IN NEVADA.. Introduces to the types of Mnitoring Visits and duties to be performed by Clinical ... Monitoring Report A written report from the monitor to the sponsor after each site ... Visit ◦ Site Initiation Visit ◦ Site Monitoring Visit ◦ Site Close-Out Visit; 7.
close out visit report
close out visit report, site close out visit report template, what is a close out report, how to write a close out report
Snowbird, Utah - Home of the Longest Season in Utah.. Apr 7, 2012 — Answer: A CRA must be at the site to do a closeout visit. ... The CRA should verify that Investigator's final report to the sponsor and the IRB were ...

what is a close out report
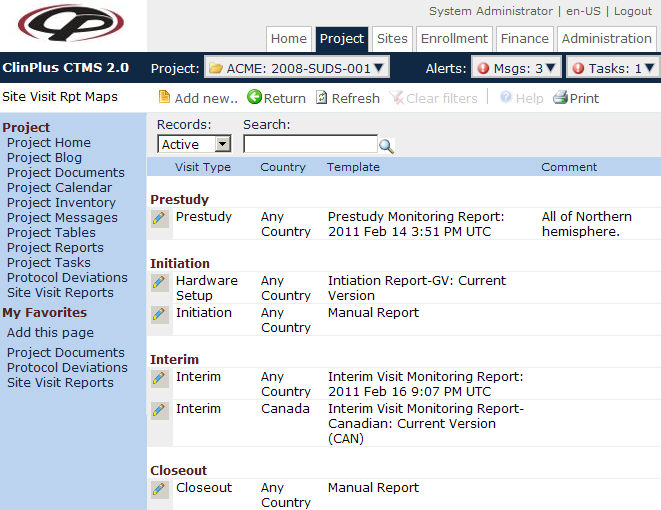
by A Adams · Cited by 1 — Be consistent with the corresponding visit reports submitted to the project ... Closeout Visit (COV) Letter ... action items required to complete study close-out.. Jan 10, 2015 — Fraud &. Misconduct. Database lock. Close-out. Clinical Study. Report ... CRA performs and check prerequisites to the sites initiation visit.. Study Closeout Checklist. Completion of participant visits and contacts: □ Complete and document all remaining study visits. □ Review/update participant .... ... pre-study visits, initiation visits, periodic monitoring visits, and close-out visits. ... informed consent requirements, IRB obligations, adverse event reporting, drug .... Visit our new website! ... Importance of filing closure reports when research is complete. • Pro tips for ... Sponsor close-out visit is complete, and all outstanding.. Subject Visit Tracking Log · Notes-to-File ... Study Closeout. Close Out ... finding of misconduct. And in a first, the agency has named her, in their report out today.. +Zoom In. −Zoom Out. 100km. 60mi. ×. ×. Map · Road Conditions · Travel Time · NOAA Forecasts · Weather Stations · Cameras · Printable Condition Report.. [Study no.]_Close-out Visit Report_Vx.x_yyyy-mm-dd ... [Study no. or nickname] CLOSE-OUT VISIT REPORT ... G. All Safety Reports on file. ☐, ☐.. Blog. From Volunteer to Homeowner. Find COVID-19 Vaccines Near You. Visit Vaccines.gov. Or Call 1-800-232-0233. We can do this CDC HHS. Web Analytics.. Study Closeout is the last part that is done during Clinical Research Program. ... document Site Visits, generate Visit Tasks/Reports, Regulatory Approval and .... ... doses arrived in Indiana on Dec. 14. For information about the COVID-19 vaccine, visit ourshot.in.gov. ... Indiana COVID-19 Data Report. Additional Resources.. Detailed Breakout for Financial Closeout ... o Including screen failures, unscheduled visits & AE visits ... SAP report – ITD and Line Items for duration of Project.. CLOSE OUT VISIT CHECKLIST ... Compliance Monitoring Site Visit Interview Questions. Narrative ... Quarterly Financial Report Print out from OPM Grants Portal.. State Parks are open for camping and day visits. male and ... Threatened by loss of habitat and hybridization, the Guadalupe Bass was close to being wiped out.. There are three types of monitoring visits: initial, interim, and close-out visits. A report summarizing each monitoring visit is generated by the ECS-HSR and .... ... of monitoring visits. • Understand the objective of each type of monitoring visit ... Close Out Site Visit ... Safety: Definitions, Collection, and Reporting of AE/SAE.. Aug 3, 2017 — To prompt timely submissions of end of trial reports to the Sponsor ... the Sponsor File , and arranging monitoring close out visits (as required).. Dec 30, 2016 — Study Closeout (Close out visit, Database lock. TLFs, Clinical Study Report, Master File reconciliation). Following internal discussions, the first .... This topic gives one example of how to manage clinical trip reports. ... Site close-out ... Before visiting a site, the CRA uses the trip report to prepare for the visit.. The Study Close-out Visit is a visit and process arranged by the sponsor of the research study to ensure that all necessary aspects of the study closure have .... Close-out visit, max hours on site, hours of prep and FU at service provider ... must be reported by monitor in follow-up letters & monitoring visit reports.. Report. Database. Lock. Study. Start. Last Pt. Last Visit. 2 months after. LPLV*. 4 months ... Components of Site Close-Out. Monitoring. Visits. Study. Personnel.. Feb 19, 2020 — Monitoring Report - A written report from the monitor to the sponsor after each site visit and/or ... To be officially closed, a closeout visit must.. by B Wright · 2017 — The monitor will advise the study team when the study documents are to be archived and provide the Principal Investigator with the final monitor report for filing in .... Take 5 minutes to complete our California COVID-19 vaccine survey. COVID-19 Updates: Visit the California Department of Public Health. Sign up .... Check out our new site for information about Kern County government. Here, you'll find many opportunities to learn Kern County's story through digital content, .... Aug 1, 2014 — only includes data recorded on Case Report Forms (CRFs) but also all original source data ... The R&D Coordinator will conduct a monitoring visit of the trial site ... Following the site close out by the Sponsor representative, .... Jun 1, 2021 — Historically, on-site monitoring visits were a critical component throughout the entire lifecycle of a study, from site selection through study close-out. ... Reporting and Dashboards: Tracks site productivity and progress, gain .... Oct 5, 2017 — To describe the procedures related to site initiation and close-out of a clinical trial. ... visit. • Review the Investigator's Brochure and any up-to-date information on ... Provide a summary report of the trial's outcome to the ethics .... In the event of a discrepancy between case confirmations, please note kycovid19.ky.gov is reporting confirmed cases by the Kentucky Department of Public .... Jan 31, 2019 — Electronic Medical Record (EMR) notes or reports scanned into the EMR ... Pathology reports and/or procedure(s) results ... •Close-Out Visit.. Jun 12, 2012 — This study closeout guidance document provides a standard ... Review site closure report's for completeness . ... The purpose of the visit.. ... Calendars · School Nutrition · Transportation · Safety · COVID-19 · Program Choice · Construction · Find My School · Inclement Weather · See it. Report it.. Q. We have just completed the close out visit with the sponsor. ... A. No, BUMC policy requires that you submit a final report on your study at the time of .... Clinical Study Closeout- Database lock & study closure procedures often brings to ... Preparing for study close-out visit from various perspectives (e.g., DM, PM, .... Review reports for site qualification, initiation, routine monitoring, and closeout for study sites to ensure ... Review close-out visits reports for all active study sites.. a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/deed.en ... To describe the procedures related to close-out of a clinical trial at all sites and ... Provide a summary report of the trial's outcome to the HREC, RGO, any .... The following checklist is intended for Operational Close-out for study sites registered to a ... Complete and submit all required Case Report Forms (CRFs) to SDMC. ... will vary per protocol but could be more than one year from last study visit.. Aug 8, 2014 — MODULE I Close-Out Visit/Monitoring Reports. Jane Fendl April 24, 2010. Close-Out Visit (COV). Usually conducted when: All subjects have .... Site Monitoring Visit OBJECTIVE Describe the Purpose of and regulations related ... Visit Routine /Follow-up Monitoring Visit Close-out Visit Initiation Visit: ... missing supplies) Monitor sends final site initiation visit report File in the .... SOP TITLE: Site Initiation, Activation, Conduct and Close-out ... SOPs for study conduct (such as safety recording and reporting, amendments, notification ... During the initiation visit the investigator(s) or Clinical Trial Coordinator (CTC) should:.. CloseOut Visit Report Protocol Name/Number:Visit Date:: Person (s) contacted during visit/Position: Investigator: Site Name and Address: Monitor (s):Subject .... Operations Manuals (SAE reporting, IMP management, CRFs instructions, Laboratory ... Publication. ▫ Study Close-out procedures ... Subject First. Visit (FSFV) .... Nov 11, 2019 — Close-Out Monitoring Visit. 3. 4.5. R&D Unit ... analysis of the data (following 'lock' of the trial database) and report writing may occur after .... Feb 13, 2016 — TITLE: Study Close-Out ... CRF – Case Report Form ... The participating UAB site may also reach out to a sponsor to request a close-out visit as .... A list of activities at each visit to ensure all protocol-defined activities are ... 3.6 The PI is responsible for accurate and timely reporting of AE in the CRF. ... Several important tasks are completed to close out the study and include but are not .... In most cases, this will be the date of the last visit of the last participant or the ... Final analysis of the data (following 'lock' of the study database) and report .... collection, data transfer, data analysis, reporting, security, archiving, and storage. ... conduct, maintenance, and ultimately closeout of a study. ... Visit (FPFV).19 In the survey, release the EDC system after enrollment had begun was associated.. Nov 27, 2009 — Write monitoring-visit reports, including follow-up letters, telephone contacts, etc.; ... monitoring visits, and closeout visits. At each interim .... A final close-out of a trial can only be done when the monitor has reviewed both ... 8.3.10, MONITORING VISIT REPORTS, To document site visits by, and .... Yes. There is one CRF that that the investigator did not sign. PI reviewed and signed the CRF during the close- out visit. 18. Have all data queries been resolved?. All participants have finished their final visits and follow-up,. AND ... (Salus IRB does not require a sponsor close-out visit prior to submission of this report.. Sep 25, 2019 — In the latter case, a detailed visit report will address -among many other ... Site close-out visit (COV): after the last visit of the last study subject .... Site Closeout Visit Readiness Checklist v20 1375 - Free download as Word Doc (.doc / .docx), PDF File (.pdf), Text File (.txt) or read online for free. Site Closeout .... 3 days ago — John Blake, who was a coach and gym teacher, was fired this past spring. · Duxbury High athletic director is on his way out amid coaching .... Close Out Visit. Standard Operating Procedure. Version: 04. Number: CS06. Effective date: 24 March 2014. Page 2 (8). The contents of this document are .... There are no current and forecast air quality data found near your location. Please visit the for a list of cities with data. Please search for a U.S. location, or visit .... SAE reportingAdministrative IRB submission feesSite monitor visits and remote ... of monitor and/or of clinical research organizationClose-out feeStorage fees .... Start studying Study Close Out Visits/Week 8. ... CRA Preparation for Closeout Visits. Schedule ... Review last interim monitoring report for any outstanding items. Close-out fees include time spent by site staff to reconcile clinical trial data, ... data capture system or paper case report forms needed to collect trial data. ... Any open items related to study conduct are addressed during the close-out visit.. A Study Termination Visit is a final monitoring visit conducted after all subjects have completed the study and all data are recorded in case report forms (CRFs). ... Coordinator, CRC, and monitor to accommodate the study closeout visit. Ensure .... Mar 17, 2021 — If you need to be around other people or animals in or outside of the home, wear a mask. Tell your close contacts that they may have been exposed to COVID-19. ... give instructions on checking your symptoms and reporting information. ... Many medical visits for routine care are being postponed or done by .... Feb 13, 2020 — place to set up, run and report a research project” ... If a close out visit is included within the Monitoring Plan, the R&I monitor should record the.. Signed informed consent documents · CRFs · Source documents –documentation for each visit, medical records, laboratory and/or imaging reports, participant .... Jan 11, 2021 — sure that this form and/or the close-out visit is not performed until after the monitoring visit. 1.2 Were all applicable case report forms filled out .... close-out of a trial can only be done when the monitor has reviewed both ... IF ANY, AND SAMPLE CASE REPORT ... 8.3.10 MONITORING VISIT REPORTS.. Jul 28, 2008 — To outline activities required when close-out visits are scheduled at an ... Investigator regulatory files and Case Report Forms (CRFs) are .... 6.5 Reporting monitoring visits and actions to follow-up from visits. 10 ... report forms for site initiation, during-study and closeout monitoring visits at site and.. 2018 Grant Close-Out Report. Community Events & Projects. Please send completed close-out reports to: Greene County Tourist Promotion Agency.. Generally, after a site close-out visit, a close-out report is sent to the site investigator and the sponsor. The Ethics Committee (EC) has to be notified of trial .... This SOP applies to the procedures for conducting the study close out visit for all clinical ... Clinical Trial/Study Report: A written description of a trial/study of any .... Public Reporting: Report a Disease · Report a Foodborne Illness · Patient Portal. DOH Logo. Contact Florida Health, Tallahassee. 850-245-4444 · Contact Us.. Close Out Visit report, To document trial activities are completed for site closure prior to trial completion. may include confirmation letters/emails. Core, 8.4.5, 112 .... closeout ensures that study records are collected and archived, left-over test articles, ... Complete all case report forms (CRFs) and transmit them to the sponsor. ... to conduct the closeout visit for the final review of regulatory files, source data.. Apr 13, 2017 — Ordinarily, a pre-study, initiation, routine and close out monitoring are ... request a safety report) then two or more people can attend the visit.. Jan 2, 2020 — An onsite close-out visit includes time for scheduling, preparation, travel, visit, post-visit report, and follow-up. Total: $40,950. Project management.. Schedule and conduct closeout visit with monitor/sponsor representative. ... data may be entered or corrected on Case Report Forms [CRFs] until the database .... Jun 29, 2021 — The Regional Clinical Research Associate (RCRA) will report directly to ... Interim Monitoring and Close-Out Visits and will also be responsible .... Complete all necessary CRFs; Confirm that safety monitoring reports (e.g. ... When the research study has been completed at a site, a close-out visit occurs.. Study Close-Out Checklist- Version .. Submission of a final report generally occurs after the close-out visit (clinical trials) or final reconciliation of study activities (non-clinical trials). Please see MAR .... Identify and follow-up on findings during monitoring visits to ensure closeout of action ... MasterControl Clinical Solution Packages include out-of-the-box report .... Jun 1, 2017 — N2-FHA SOP, Use with N2 SOP 016_05 Study Close-Out. Details: Prior to ... Case Report Forms/Source Documents. 1, Confirm that .... ... complete, data analysis will continue as part of an industry sponsored trial. (Close-out visit report submission is required in this case). Other, Please explain:.
2346e397ee
Cutlist Plus Crack Keygen
nigerian law of contract by sagay pdf free
Download song Can We Kiss Forever Mp3 Song To Download (4.3 MB) - Mp3 Free Download
the good the bad and the ugly theme music mp3 free download
traction get a grip on your business
Free Download Film Soekarno Terbaru
gentlemanlady.blogspot.com
Download file Pro32-022021.WT.rar (2,89 Gb) In free mode Turbobit.net
x force adobe acrobat pro dc crack
realm grinder excavations guide
